MDMA for PTSD
Plain-language summary
This page summarizes what randomized clinical trials currently suggest about MDMA-assisted therapy for PTSD in simple terms.
- What we asked: Do people with PTSD improve more after MDMA-assisted therapy than after a comparison condition (like a placebo or low dose), when both are paired with psychological support?
- Who was studied: Adults with elevated PTSD symptoms. All trials were conducted by the Multidisciplinary Association for Psychedelic Studies (MAPS) and its spin-off Resilient Pharmaceuticals (formerly Lykos Therapeutics).
- What was the intervention?: Trials gave MDMA at doses intended to produce a noticeable subjective experience, alongside structured psychotherapy before, during, and after dosing sessions. Participants received between two and three dosing sessions.
- What we found: Across 6 randomized studies including 286 participants, MDMA was associated with larger improvements in PTSD scores than comparison conditions. People receiving MDMA were also more likely to meet definitions of treatment response and remission. More dosing sessions and a higher cumulative dose were associated with larger effects.
- How confident are we?: We assigned an overall low certainty rating using the GRADE approach. Functional unblinding (participants and therapists guessing group assignment due to MDMA’s psychoactive effects) and the high rates of prior MDMA use among participants are key sources of concern. The true treatment effect in routine clinical practice may be smaller than what has been observed in trials so far.
- What this does not mean: This is not medical advice. MDMA is not approved to treat PTSD. The FDA denied a new drug application for MDMA-assisted therapy in 2024, citing concerns about safety assessment, durability of effects, and potential expectancy bias. Most trials were small, carefully selected populations and may not generalize to broader treatment-seeking populations.
- Safety note: This page focuses on whether PTSD symptoms improved. Adverse effects and safety were not the primary focus here and will be considered in a future analysis.
- We will keep this updated: This is a living review. As new randomized controlled trials are published, we update our analyses and figures. For methods and full details, see the reproducibility guide and search strategy. An interactive overview is available on our dashboard.
About this page
Welcome to the landing page for our living systematic review and meta-analysis on MDMA-assisted therapy for PTSD (pre-registered here)! Follow the links in our documentation section for relevant information including our code-walkthrough (reproducibility guide). Here we provide an overview on our latest results, currently up-to-date with our most recent preprint.
Eligibility Criteria
We included randomized controlled clinical trials published in English in peer-reviewed journals comparing MDMA for PTSD with a comparator in adult (>18 years old) populations. In order to meet these criteria, the study population needed to include individuals with elevated PTSD symptoms; studies with only healthy participants were not considered. Eligible interventions included any dose and formulation of MDMA or other prodrugs of MDMA intended to produce an alteration of subjective experience in the patient, with or without the conjunctive use of therapy (e.g., microdosing studies were not included). Eligible comparators included any form of placebo with or without the conjunctive use of therapy, including lower doses of the intervention drug and any dose of other psychotropics intended to improve blinding (without known therapeutic efficacy for PTSD). Eligible comparators also included control for spontaneous improvement via waitlist or usual care. Additionally, for studies that used a crossover design, data from pre-crossover timepoints was required to account for potential carry-over effects of an MDMA dose.
Study selection
We identified 1,586 reports from our searches. Of these, 26 passed initial title and abstract screening and were reviewed as full-text articles. Six studies (286 participants) met inclusion criteria for our primary model.
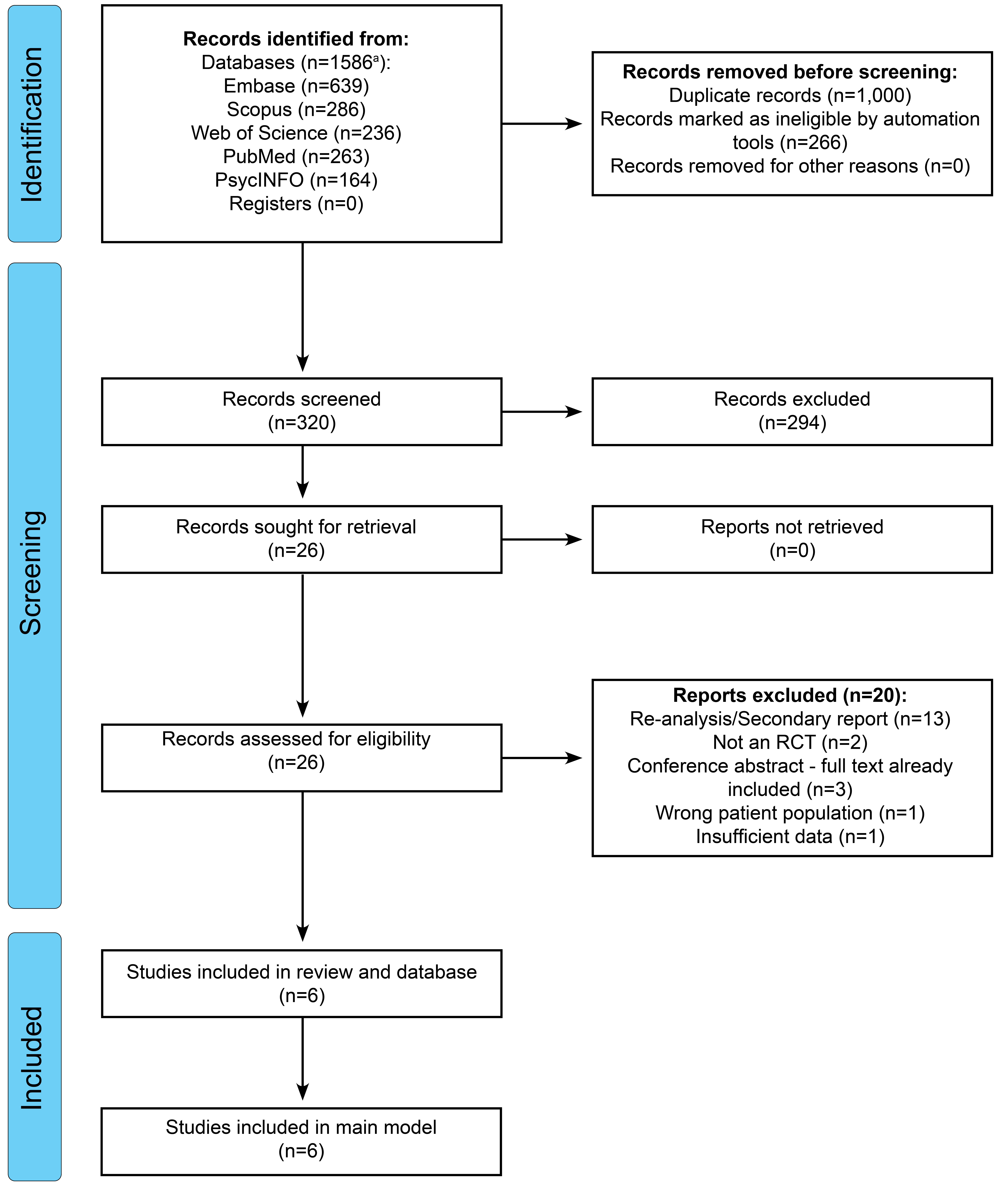 Figure 1: PRISMA flow diagram showing study selection process
Figure 1: PRISMA flow diagram showing study selection process
Study Characteristics
Our database consists of 61 effect sizes generated from 6 included studies, covering continuous and dichotomous measures of PTSD symptoms across timepoints (from four days after the first dose out to 12 months post-baseline). All studies included a psychotherapy or psychological support component before, during, and after MDMA dosing. See Table 1 for detailed study characteristics.
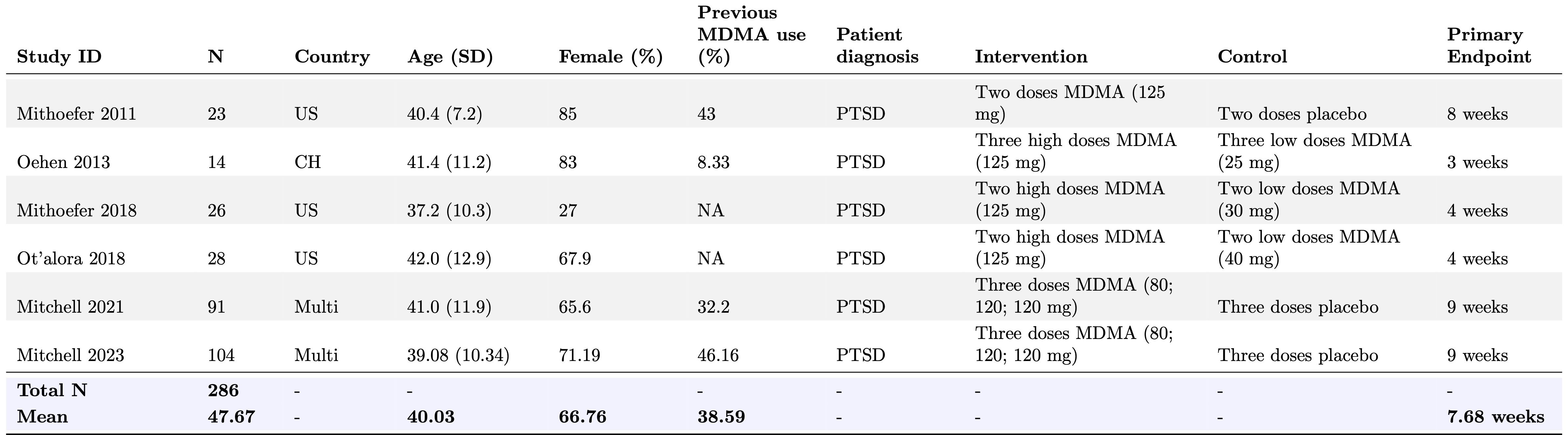 Table 1: Summary of RCTs on MDMA-assisted therapy for PTSD.
N given is the number of participants randomized. Study endpoints are the reported primary endpoints for each study. The MDMA-AT protocol typically involves preparatory psychotherapy sessions, 2–3 dosing sessions lasting 8–10 hours spaced approximately one month apart, and integration psychotherapy sessions following each dosing session.
Table 1: Summary of RCTs on MDMA-assisted therapy for PTSD.
N given is the number of participants randomized. Study endpoints are the reported primary endpoints for each study. The MDMA-AT protocol typically involves preparatory psychotherapy sessions, 2–3 dosing sessions lasting 8–10 hours spaced approximately one month apart, and integration psychotherapy sessions following each dosing session.
Risk of Bias
We assessed study bias using Cochrane’s Risk of Bias 2.0 tool, which is the standard approach for bias assessment in randomized controlled trials. We focused our assessment specifically on primary outcome variables. If information pertinent to assessment of risk of bias was not available, authors were contacted via email.
The RoB 2.0 tool examines five potential bias sources: randomization procedures, deviations from the intended protocol, missing outcome data, outcome measurement, and selective reporting. Each domain contains several questions rated on a four-level scale ranging from ‘yes’ to ‘no’, in addition to an option for insufficient information. Domain-level and overall study bias ratings are classified as low, medium, or high according to a predetermined algorithm. While this algorithm guides assessment, evaluators may justifiably override automated bias determinations when specific concerns warrant greater or lesser emphasis than the algorithm suggests.
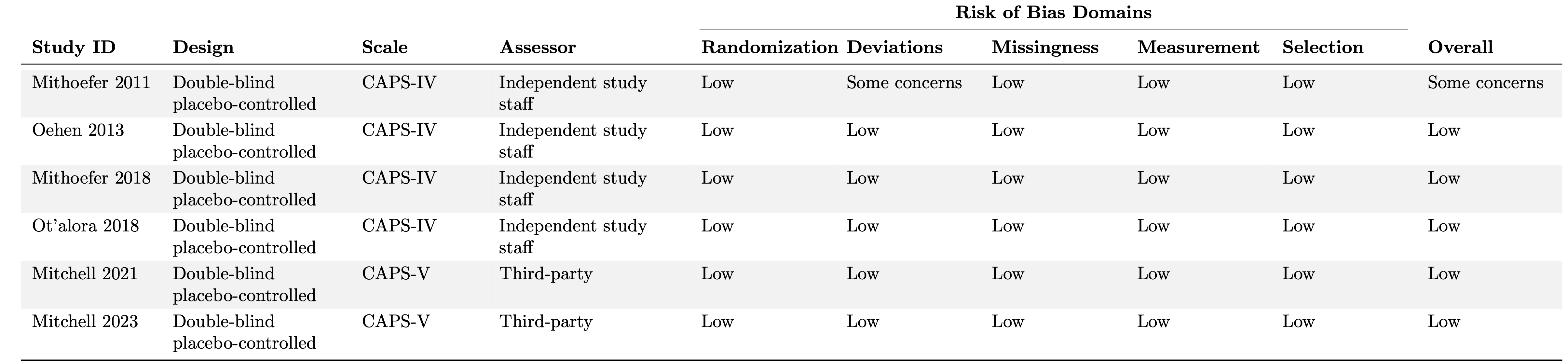 Table 2: Additional study characteristics and risk of bias assessments using Cochrane’s Risk of Bias 2.0 tool domains.
Randomization = bias due to randomization process;
Deviations = bias due to deviations from intended interventions;
Missingness = bias due to missing outcome data;
Measurement = bias due to measurement of the outcome;
Selection = bias due to selection of the reported results;
Overall = overall risk of bias.
Table 2: Additional study characteristics and risk of bias assessments using Cochrane’s Risk of Bias 2.0 tool domains.
Randomization = bias due to randomization process;
Deviations = bias due to deviations from intended interventions;
Missingness = bias due to missing outcome data;
Measurement = bias due to measurement of the outcome;
Selection = bias due to selection of the reported results;
Overall = overall risk of bias.
Overall, one study had some concerns (Mithoefer 2011) and five studies were deemed to have an overall low risk of bias (Table 2). Mithoefer 2011 had some concerns due to what appears to be a per-protocol study design, where dropouts were replaced with new enrollees. According to Cochrane guidelines, per-protocol study designs are not an appropriate analysis to estimate the effect of assignment to intervention. Although none of the studies had a high risk of bias, these assessments do not fully account for potential bias from functional unblinding or expectancy effects.
Results
MDMA treatment significantly reduces PTSD symptoms compared with control conditions
The analysis on continuous outcomes for the six studies included in the primary model showed a statistically significant reduction in PTSD scores after MDMA treatment compared with control conditions (Figure 2; Hedges’ g = -0.71 [-0.95; -0.47], p < 0.001, k = 6, n = 242), with low between-study heterogeneity (tau2 = 0.00 [0.00; 0.38], I2 = 0.0% [0.0%; 74.6%]).
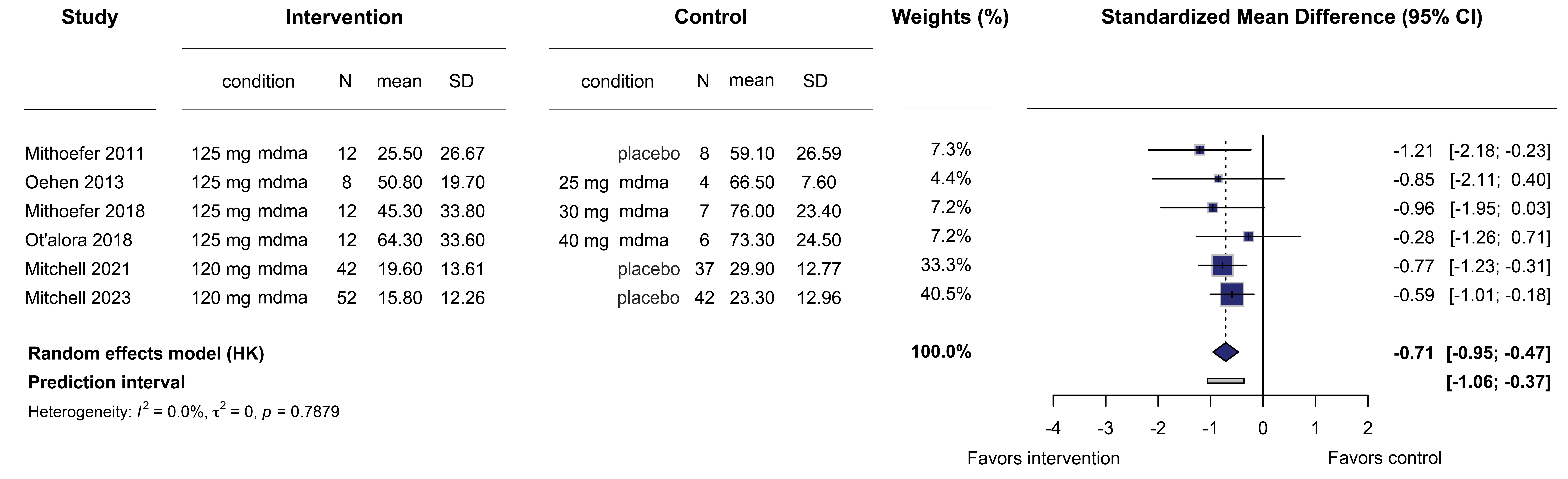 Figure 2: Meta-analysis on continuous outcome variables.
Boxes represent the standardized mean difference (Hedges’ g) for each study, and the lines extending from the box represent the 95% confidence interval around each effect size, while the size of each box is proportional to its weight.
The diamond at the bottom represents the pooled effect size (meta-analytic mean).
The gray line at the bottom represents the prediction interval of the expected range of true effects in a new study. HK = Knapp-Hartung adjustment.
Figure 2: Meta-analysis on continuous outcome variables.
Boxes represent the standardized mean difference (Hedges’ g) for each study, and the lines extending from the box represent the 95% confidence interval around each effect size, while the size of each box is proportional to its weight.
The diamond at the bottom represents the pooled effect size (meta-analytic mean).
The gray line at the bottom represents the prediction interval of the expected range of true effects in a new study. HK = Knapp-Hartung adjustment.
Visual inspection of a funnel plot (SI Figure 1) revealed limited asymmetry. An Egger’s test did not find small study effects (intercept = -0.52 [-1.94; 0.91], t = -0.71, p = 0.52), although this test is underpowered given the small number of studies included in our meta-analysis (k < 10).
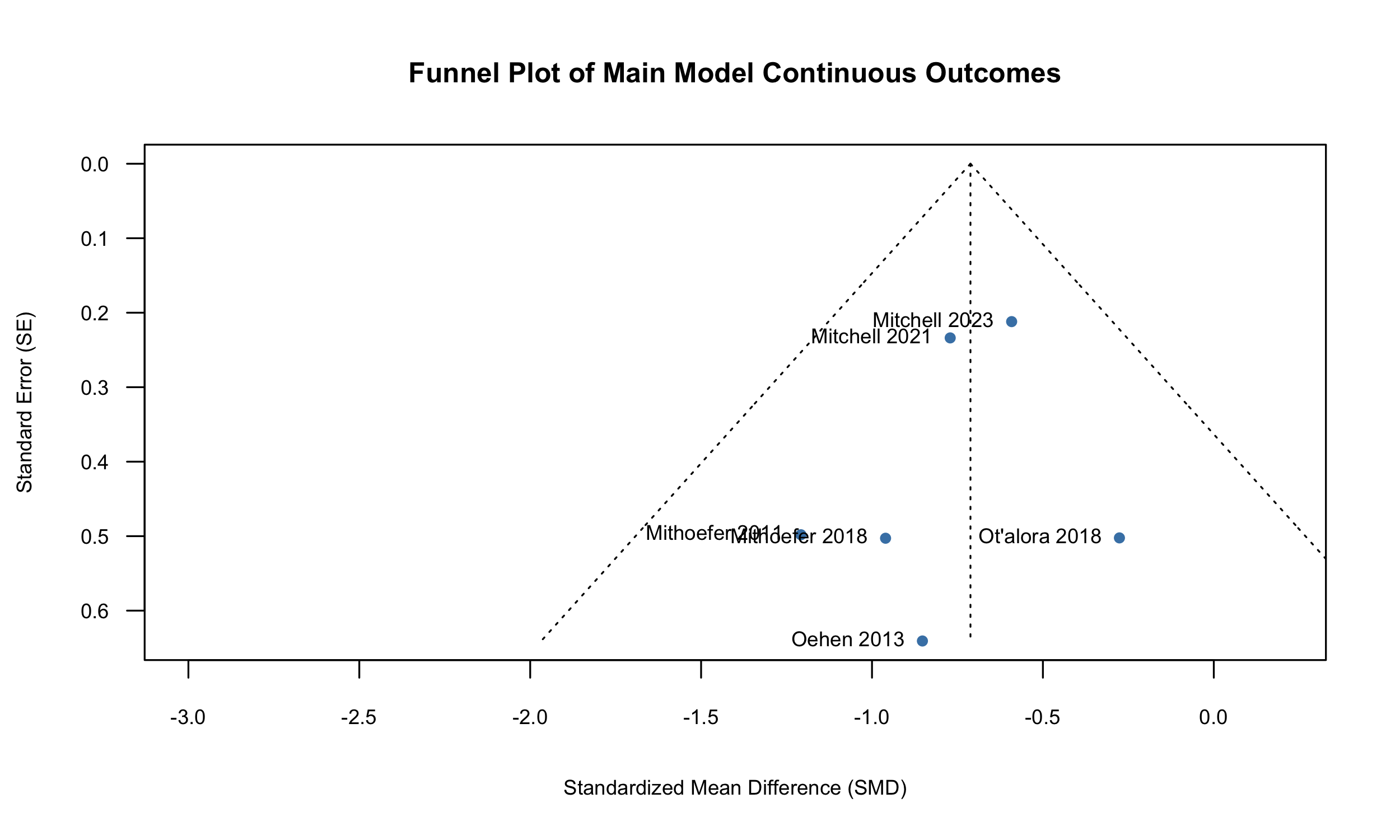 SI Figure 1: Funnel plot of 6 studies in primary meta-analytic model.
SI Figure 1: Funnel plot of 6 studies in primary meta-analytic model.
MDMA’s effects increase over dosing sessions and cumulative dose
Our three-level CHE model revealed a significant decrease in PTSD scores with MDMA compared to the control conditions consistent with our primary model (Hedges’ g = -0.60 [-0.86; -0.34], p < 0.001, k = 6, n = 286, tau2 = 0.00 [0.00; 0.23], I2 = 0.0% [0.0%; 66.0%]) (Figure 3a).
We next performed two separate meta-regressions. Adding the number of dosing sessions as a continuous predictor to our model (Figure 3b) revealed a significant effect of increased dosing sessions, such that more dosing sessions resulted in a greater between-group difference in PTSD scores (β = -0.20 Hedges’ g [-0.34; -0.06], p = 0.01). We also evaluated the cumulative MDMA dose as a continuous predictor; this revealed a significant effect of increased MDMA dose, such that a higher dose exposure resulted in a greater between-group difference in PTSD scores (β = -0.002 Hedges’ g/mg [-0.003; -0.0005], p = 0.01).
 Figure 3: Three-level CHE model results. (a) Forest plot of the overall three-level model. (b) Meta-regression of the number of dosing sessions on effect size.
Figure 3: Three-level CHE model results. (a) Forest plot of the overall three-level model. (b) Meta-regression of the number of dosing sessions on effect size.
Higher response and remission rates, nonsignificant effect on depression symptoms after MDMA treatment
We found evidence for statistically significant greater treatment response with MDMA compared with control conditions (Figure 4a; RR = 1.35 [1.10; 1.66], p = 0.016, k = 5, n = 222), with low between-study heterogeneity (tau2 = 0.00 [0.00; 1.68]; I2 = 0.0% [0.0%; 79.2%]).
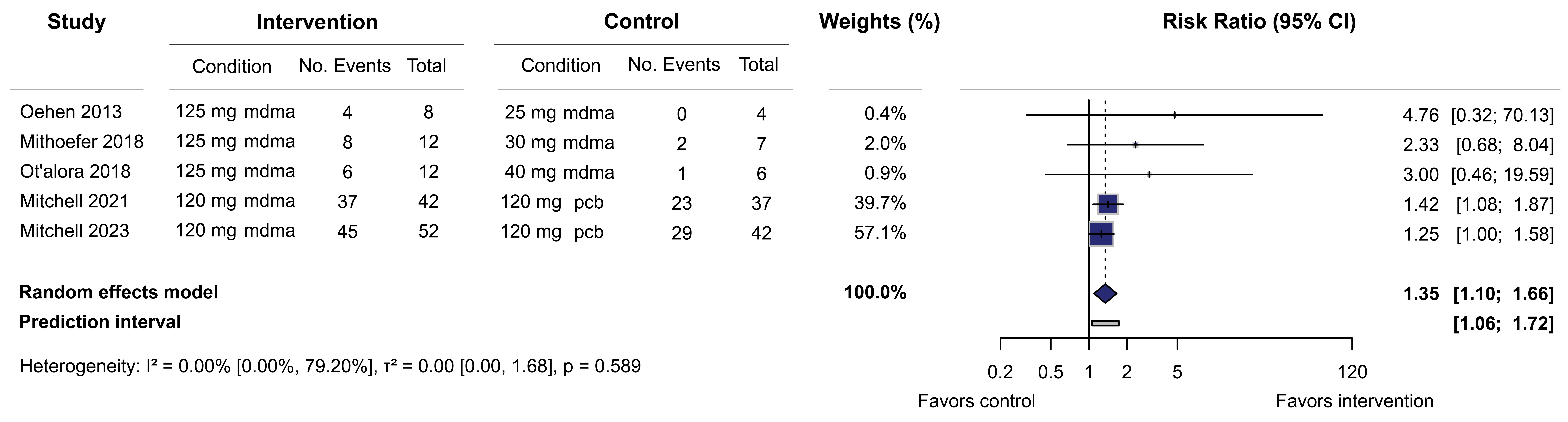 Figure 4a: Meta-analysis on response to treatment.
Boxes represent the risk ratio (RR) for each study, and the lines extending from the box represent the 95% confidence interval around each effect size, while the size of each box is proportional to its weight.
The diamond at the bottom represents the pooled effect size (meta-analytic mean).
The gray line at the bottom represents the prediction interval of the expected range of true effects in a new study.
Figure 4a: Meta-analysis on response to treatment.
Boxes represent the risk ratio (RR) for each study, and the lines extending from the box represent the 95% confidence interval around each effect size, while the size of each box is proportional to its weight.
The diamond at the bottom represents the pooled effect size (meta-analytic mean).
The gray line at the bottom represents the prediction interval of the expected range of true effects in a new study.
We also found that there were significantly higher remission rates with MDMA compared with control conditions (Figure 4b; RR = 2.25 [1.04; 4.87], p = 0.044, k = 4, n = 210), with low between-study heterogeneity (tau2 = 0.00 [0.00; 5.78]; I2 = 0.0% [0.0%; 84.7%]).
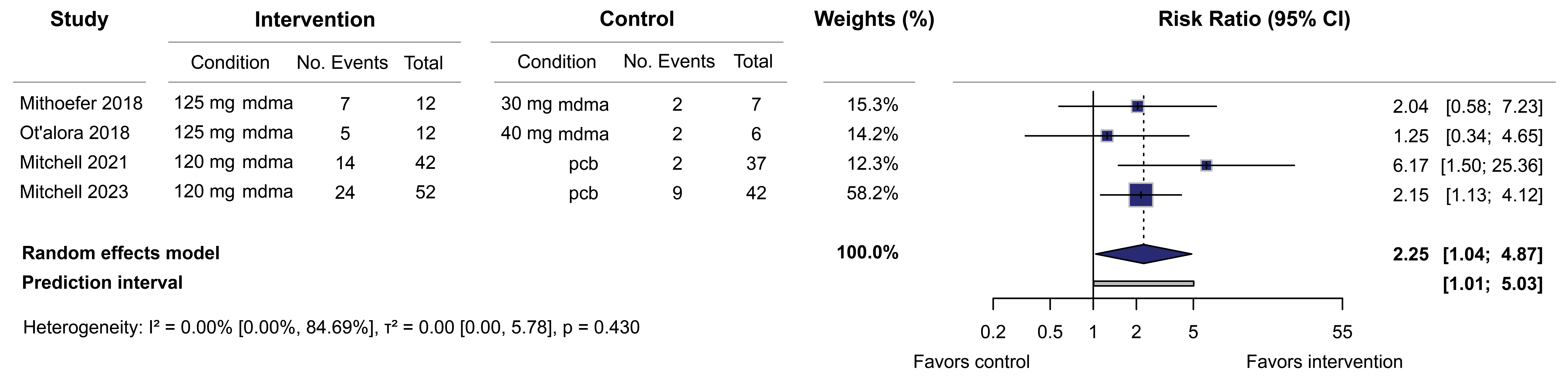 Figure 4b: Meta-analysis on remission rates.
Boxes represent the risk ratio (RR) for each study, and the lines extending from the box represent the 95% confidence interval around each effect size, while the size of each box is proportional to its weight.
The diamond at the bottom represents the pooled effect size (meta-analytic mean).
The gray line at the bottom represents the prediction interval of the expected range of true effects in a new study.
Figure 4b: Meta-analysis on remission rates.
Boxes represent the risk ratio (RR) for each study, and the lines extending from the box represent the 95% confidence interval around each effect size, while the size of each box is proportional to its weight.
The diamond at the bottom represents the pooled effect size (meta-analytic mean).
The gray line at the bottom represents the prediction interval of the expected range of true effects in a new study.
In contrast, we did not find evidence for a significant effect of MDMA on co-morbid depression symptoms (Figure 4c; Hedges’ g = -0.66 [-2.70; 1.39], p = 0.30), with a low number of studies (k = 3, n = 118) and high heterogeneity (tau2 = 0.44 [0.00; 29.60], I2 = 68.0% [0.0%; 90.7%]).
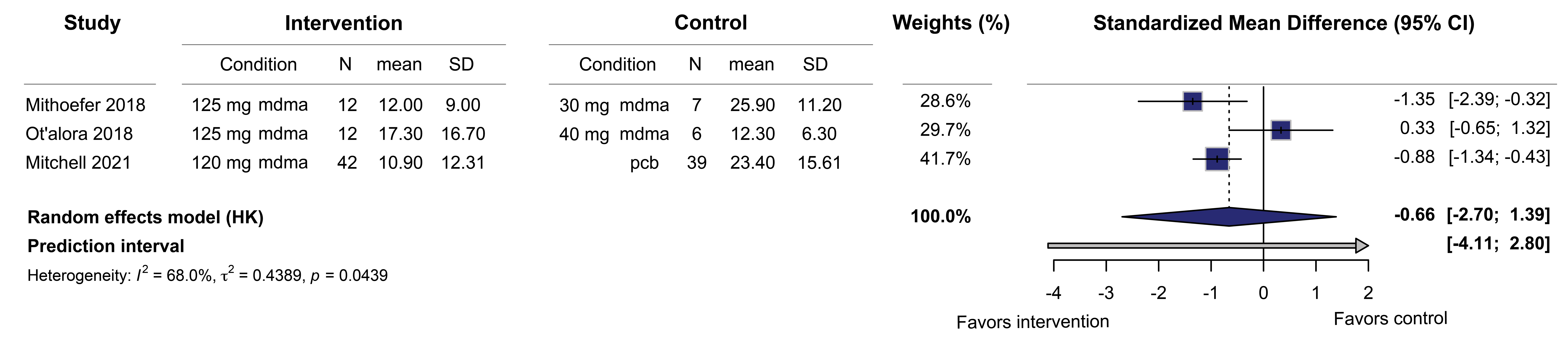 Figure 4c: Meta-analysis on co-morbid depression symptoms in PTSD.
Figure 4c: Meta-analysis on co-morbid depression symptoms in PTSD.
Sensitivity analyses
To assess the robustness of our primary findings, we performed the following sensitivity analyses:
- Alternate dosing in three-arm trials: Given that Mithoefer 2018 and Ot’alora G 2018 employed a three-arm design comparing MDMA at high and medium doses against a low-dose control, we conducted a sensitivity analysis substituting the medium dose intervention arm for the high-dose intervention arm used in our primary analysis.
- Fixed effect models: We ran fixed/common-effect models as sensitivity analyses to compare to random effects models. The fixed effect model assumes that the between-study variance (tau2) is 0, such that all studies share a common true effect size. For our continuous model, we used a standard inverse-variance weighting fixed-effect model on standardized mean differences (Hedges’ g). For our dichotomous models, we used a standard inverse-variance weighting fixed-effect model on the log risk ratio.
- Three-level model within-study correlation coefficient sweep: Our three-level CHE model assumed a constant within-study correlation coefficient (ρ) of 0.6. To test the sensitivity of our results against this approximation, we recalculated Hedges’ g as a function of ρ from 0 to 1 in 0.1 increments.
- Bayesian meta-analysis: We replicated our primary meta-analysis on continuous outcomes using a Bayesian implementation. We used “weakly informative” prior distributions for both the main effect and the heterogeneity parameter tau that have been recommended by prior work. The main effect prior was a normal distribution centered around 0, with a standard deviation of 1, while the tau prior was a half-normal distribution with a standard deviation of 0.5.
Sensitivity analyses provide convergent results
We performed a series of sensitivity analyses that supported our primary results. First, our model using medium-dose arms in place of the high-dose arms for three-arm trials showed a significant and comparable effect size (SI Figure 2; Hedges’ g = -0.75 [-1.19; -0.30], p = 0.008, k = 6, n = 234, tau2 = 5.31 x 10-6 [0.00; 3.11], I2 = 37.2% [0.0%; 75.0%]).
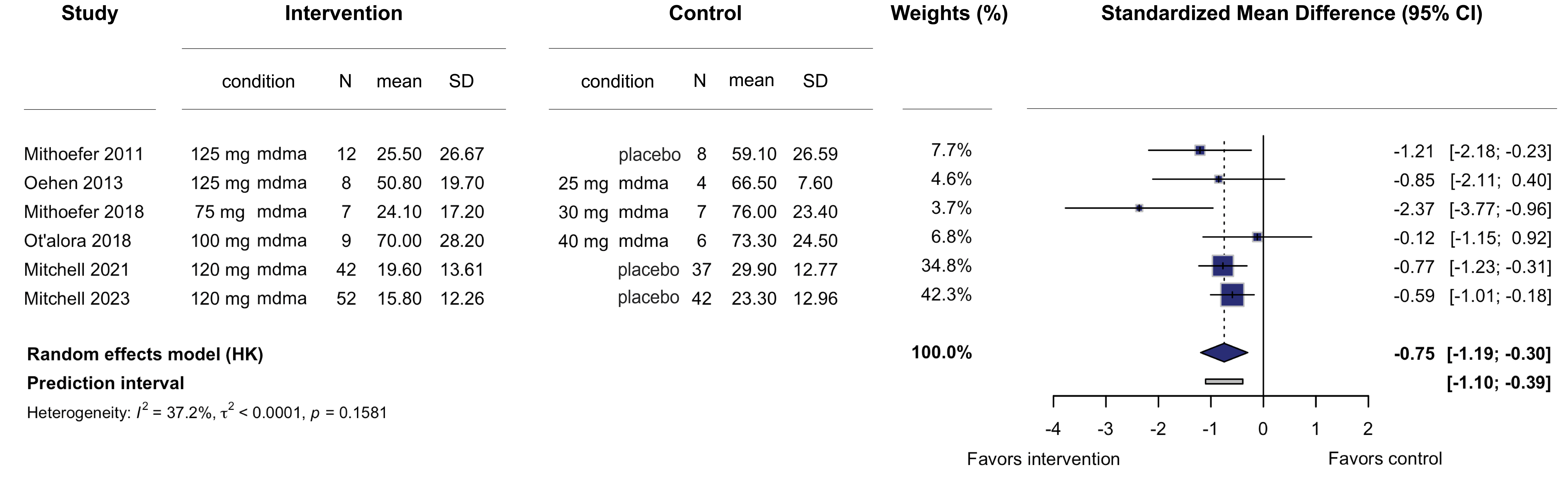 SI Figure 2: Forest plot of 6 studies using medium-dose arms in three-arm trials. Boxes represent the standardized mean difference (Hedges’ g) for each study, and the lines extending from the box represent the 95% confidence interval around each effect size, while the size of each box is proportional to its weight. The diamond at the bottom represents the pooled effect size (meta-analytic mean). The gray line at the bottom represents the prediction interval of the expected range of true effects in a new study. HK = Knapp-Hartung adjustment.
SI Figure 2: Forest plot of 6 studies using medium-dose arms in three-arm trials. Boxes represent the standardized mean difference (Hedges’ g) for each study, and the lines extending from the box represent the 95% confidence interval around each effect size, while the size of each box is proportional to its weight. The diamond at the bottom represents the pooled effect size (meta-analytic mean). The gray line at the bottom represents the prediction interval of the expected range of true effects in a new study. HK = Knapp-Hartung adjustment.
Furthermore, we replicated our primary analyses using a fixed effect model on continuous outcomes (Hedges’ g = -0.71 [-0.98; -0.45], p < 0.001, k = 6, n = 242), response outcomes (RR = 1.44 [1.20; 1.74], p < 0.001, k = 5, n = 222), and remission outcomes (RR = 2.49 [1.52; 4.09], p < 0.001, k = 4, n = 210).
Our Bayesian analysis revealed a Hedges’ g posterior distribution centered at -0.70 [-1.05; -0.36] (SI Figure 3).
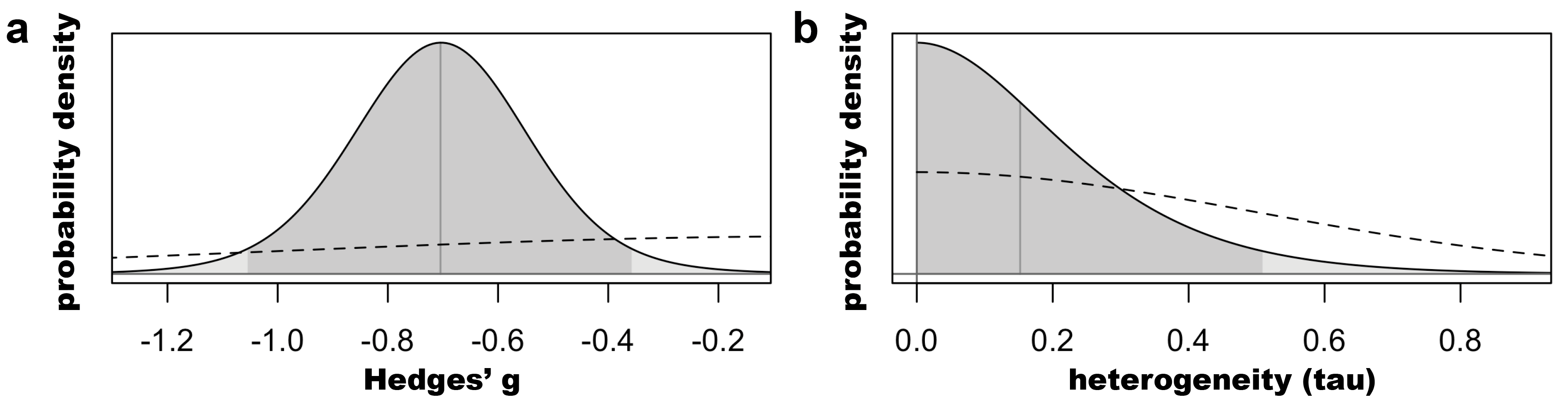 SI Figure 3: Prior (dashed) and posterior (solid) distributions for (a) the pooled effect size (Hedges’ *g) and (b) the heterogeneity estimate (tau). Dark shading represents the 95% probability distribution.*
SI Figure 3: Prior (dashed) and posterior (solid) distributions for (a) the pooled effect size (Hedges’ *g) and (b) the heterogeneity estimate (tau). Dark shading represents the 95% probability distribution.*
Finally, our three-level CHE results were consistent across a range of within-study correlation coefficients (SI Figure 4).
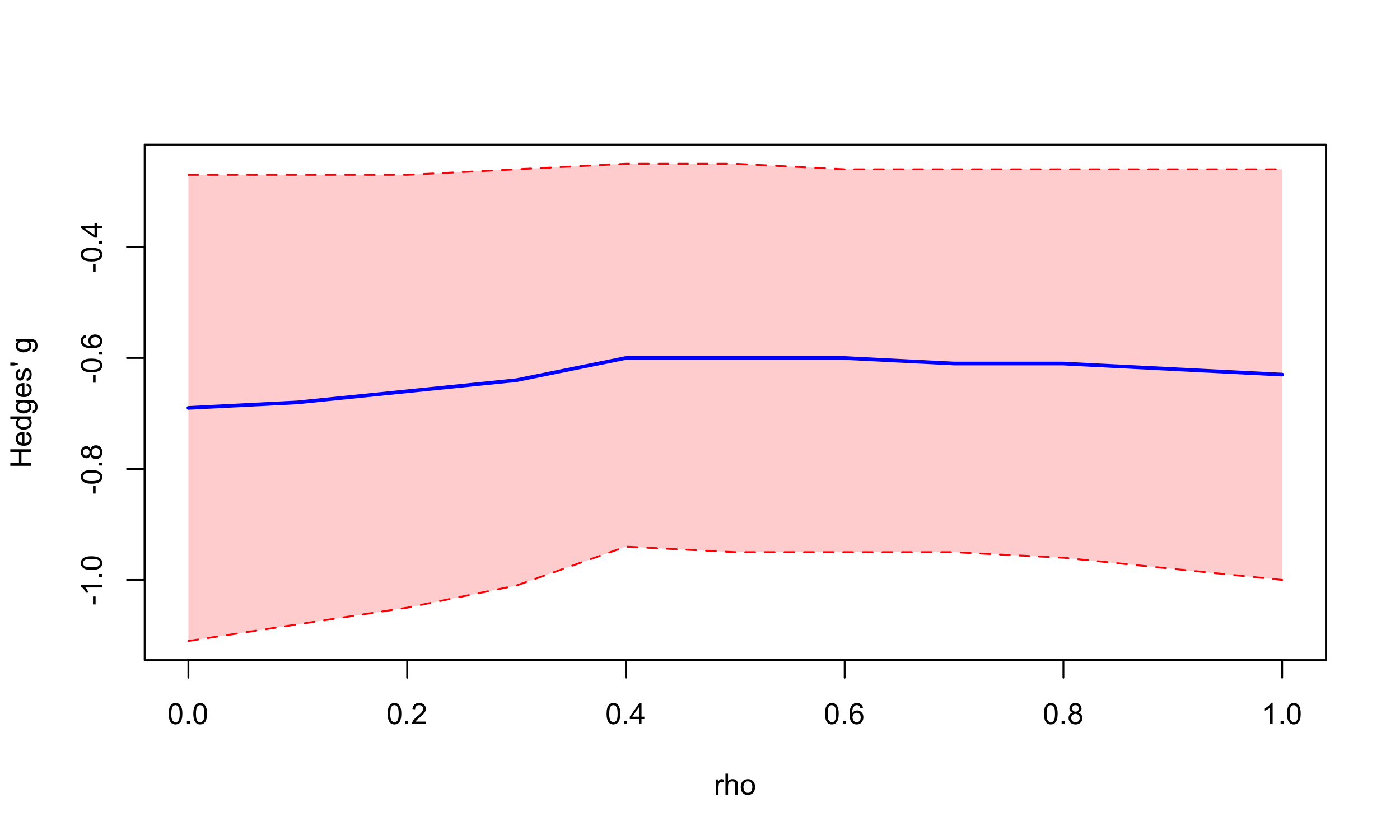 SI Figure 4: Hedges’ *g as a function of rho (ρ), the within-study correlation coefficient used in the three-level CHE model.*
SI Figure 4: Hedges’ *g as a function of rho (ρ), the within-study correlation coefficient used in the three-level CHE model.*
GRADE Certainty of Evidence
Evidence derived from randomized controlled trials begins with a high certainty rating that can then be downgraded to moderate, low, or very low depending on assessments across four domains: risk of bias, inconsistency, indirectness, and imprecision. We downgraded the certainty of evidence by two levels for indirectness. Functional unblinding, particularly in the two large phase 3 trials which used an inert placebo as comparator, presents risks for expectancy effects to drive larger between-group differences in outcomes. Further, the percentage of participants with prior MDMA use in the studies was high (mean: 39%). Taken together, these factors of indirectness may lead to smaller treatment effects in a general population under routine clinical practice and contribute to an overall GRADE rating of low certainty. No downgrades were given for risk of bias, inconsistency, or imprecision.
Conclusions
Together, synthesis of studies to date suggest that MDMA-assisted psychotherapy results in reductions in PTSD symptoms and increased response and remission in patients with PTSD. However, our GRADE rating for the certainty of this evidence is low. Additional, large controlled trials with rigorous methods are needed, as are studies examining varying study characteristics (e.g., dosing sessions, longer follow-up durations) and expanding to more representative populations. As more RCTs are published, we will regularly update our SYPRES website and dashboard in a reproducible and transparent manner. This living systematic review, in conjunction with the associated open-science resources, aims to provide a valuable and transparent resource for researchers, clinicians, policymakers, and the public.
References
-
Mithoefer, M.C., Wagner, M.T., Mithoefer, A.T., Jerome, L., Doblin, R. (2011). The safety and efficacy of {+/-}3,4-methylenedioxymethamphetamine-assisted psychotherapy in subjects with chronic, treatment-resistant posttraumatic stress disorder: the first randomized controlled pilot study. J Psychopharmacol, 25(4), 439-52. https://doi.org/10.1177/0269881110378371
Study summary via Blossom
In the first randomised controlled pilot trial, MDMA-assisted psychotherapy produced significantly greater reductions in Clinician-Administered PTSD Scale scores than placebo in 20 patients with chronic, treatment‑resistant post‑traumatic stress disorder (83% response vs 25%). There were no drug‑related serious adverse events, adverse neurocognitive effects or clinically significant blood pressure increases, suggesting MDMA may be a safe and effective adjunct to psychotherapy in refractory PTSD.
Abstract
Case reports indicate that psychiatrists administered ±3,4-methylenedioxymethamphetamine (MDMA) as a catalyst to psychotherapy before recreational use of MDMA as ‘Ecstasy’ resulted in its criminalization in 1985. Over two decades later, this study is the first completed clinical trial evaluating MDMA as a therapeutic adjunct. Twenty patients with chronic posttraumatic stress disorder, refractory to both psychotherapy and psychopharmacology, were randomly assigned to psychotherapy with concomitant active drug ( n = 12) or inactive placebo ( n = 8) administered during two 8-h experimental psychotherapy sessions. Both groups received preparatory and follow-up non-drug psychotherapy. The primary outcome measure was the Clinician-Administered PTSD Scale, administered at baseline, 4 days after each experimental session, and 2 months after the second session. Neurocognitive testing, blood pressure, and temperature monitoring were performed. After 2-month follow-up, placebo subjects were offered the option to re-enroll in the experimental procedure with open-label MDMA. Decrease in Clinician-Administered PTSD Scale scores from baseline was significantly greater for the group that received MDMA than for the placebo group at all three time points after baseline. The rate of clinical response was 10/12 (83%) in the active treatment group versus 2/8 (25%) in the placebo group. There were no drug-related serious adverse events, adverse neurocognitive effects or clinically significant blood pressure increases. MDMA-assisted psychotherapy can be administered to posttraumatic stress disorder patients without evidence of harm, and it may be useful in patients refractory to other treatments.
Enriched metadata from Blossom.
-
Oehen, P., Traber, R., Widmer, V., Schnyder, U. (2013). A randomized, controlled pilot study of MDMA (± 3,4-Methylenedioxymethamphetamine)-assisted psychotherapy for treatment of resistant, chronic Post-Traumatic Stress Disorder (PTSD). J Psychopharmacol, 27(1), 40-52. https://doi.org/10.1177/0269881112464827
Study summary via Blossom
In a randomized, double‑blind, active‑placebo controlled pilot trial of 12 patients with treatment‑resistant chronic PTSD, MDMA‑assisted psychotherapy was safely administered with no drug‑related serious adverse events. The intervention produced significant self‑reported symptom improvement (PDS, p=0.014), a non‑significant clinician‑rated CAPS reduction at end‑of‑treatment (p=0.066) that improved by 1‑year follow‑up, and three MDMA sessions were more effective than two (p=0.016).
Abstract
Psychiatrists and psychotherapists in the US (1970s to 1985) and Switzerland (1988–1993) used MDMA legally as a prescription drug, to enhance the effectiveness of psychotherapy. Early reports suggest that it is useful in treating trauma-related disorders. Recently, the first completed pilot study of MDMA-assisted psychotherapy for PTSD yielded encouraging results. Designed to test the safety and efficacy of MDMA-assisted psychotherapy in patients with treatment-resistant PTSD; our randomized, double-blind, active-placebo controlled trial enrolled 12 patients for treatment with either low-dose (25 mg, plus 12.5 mg supplemental dose) or full-dose MDMA (125 mg, plus 62.5 mg supplemental dose). MDMA was administered during three experimental sessions, interspersed with weekly non-drug-based psychotherapy sessions. Outcome measures used were the Clinician-Administered PTSD Scale (CAPS) and the Posttraumatic Diagnostic Scale (PDS). Patients were assessed at baseline, three weeks after the second and third MDMA session (end of treatment), and at the 2-month and 1-year follow-ups. We found that MDMA-assisted psychotherapy can be safely administered in a clinical setting. No drug-related serious adverse events occurred. We did not see statistically significant reductions in CAPS scores ( p = 0.066), although there was clinically and statistically significant self-reported (PDS) improvement ( p = 0.014). CAPS scores improved further at the 1-year follow-up. In addition, three MDMA sessions were more effective than two ( p = 0.016).
Enriched metadata from Blossom.
-
Mithoefer, M.C., Mithoefer, A.T., Feduccia, A.A., Jerome, L., Wagner, M., Wymer, J., Holland, J., Hamilton, S., Yazar-Klosinski, B., Emerson, A., Doblin, R. (2018). 3,4-methylenedioxymethamphetamine (MDMA)-assisted psychotherapy for post-traumatic stress disorder in military veterans, firefighters, and police officers: a randomised, double-blind, dose-response, phase 2 clinical trial. Lancet Psychiatry, 5(6), 486-497. https://doi.org/10.1016/S2215-0366(18)30135-4
Study summary via Blossom
This Phase II clinical trial (n=26) finds that MDMA-assisted psychotherapy (75-125 mg) led to significant and sustained decreases in PTSD (CAPS-IV) scores as compared to an active placebo (30 mg). At the 12-month follow-up, the average CAPS-IV score had dropped from 87 to 39 (67% no longer qualified for PTSD diagnosis).
Abstract
Background
Post-traumatic stress disorder (PTSD) is prevalent in military personnel and first responders, many of whom do not respond to currently available treatments. This study aimed to assess the efficacy and safety of 3,4-methylenedioxymethamphetamine (MDMA)-assisted psychotherapy for treating chronic PTSD in this population.
Methods
We did a randomised, double-blind, dose-response, phase 2 trial at an outpatient psychiatric clinic in the USA. We included service personnel who were 18 years or older, with chronic PTSD duration of 6 months or more, and who had a Clinician-Administered PTSD Scale (CAPS-IV) total score of 50 or greater. Using a web-based randomisation system, we randomly assigned participants (1:1:2) to three different dose groups of MDMA plus psychotherapy: 30 mg (active control), 75 mg, or 125 mg. We masked investigators, independent outcome raters, and participants until after the primary endpoint. MDMA was administered orally in two 8-h sessions with concomitant manualised psychotherapy. The primary outcome was mean change in CAPS-IV total score from baseline to 1 month after the second experimental session. Participants in the 30 mg and 75 mg groups subsequently underwent three 100-125 mg MDMA-assisted psychotherapy sessions in an open-label crossover, and all participants were assessed 12 months after the last MDMA session. Safety was monitored through adverse events, spontaneously reported expected reactions, vital signs, and suicidal ideation and behaviour.
Findings
Between Nov 10, 2010, and Jan 29, 2015, 26 veterans and first responders met eligibility criteria and were randomly assigned to receive 30 mg (n=7), 75 mg (n=7), or 125 mg (n=12) of MDMA plus psychotherapy. At the primary endpoint, the 75 mg and 125 mg groups had significantly greater decreases in PTSD symptom severity (mean change CAPS-IV total scores of −58·3 [SD 9·8] and −44·3 [28·7]; p=0·001) than the 30 mg group (−11·4 [12·7]). Compared with the 30 mg group, Cohen's d effect sizes were large: 2·8 (95% CI 1·19-4·39) for the 75 mg group and 1·1 (0·04-2·08) for the 125 mg group. In the open-label crossover with full-dose MDMA (100-125 mg), PTSD symptom severity significantly decreased in the group that had previously received 30 mg (p=0·01), whereas no further significant decreases were observed in the group that previously achieved a large response after 75 mg doses in the blinded segment (p=0·81). PTSD symptoms were significantly reduced at the 12-month follow-up compared with baseline after all groups had full-dose MDMA (mean CAPS-IV total score of 38·8 [SD 28·1] vs 87·1 [16·1]; p<0·0001). 85 adverse events were reported by 20 participants. Of these adverse events, four (5%) were serious: three were deemed unrelated and one possibly related to study drug treatment.
Interpretation
Active doses (75 mg and 125 mg) of MDMA with adjunctive psychotherapy in a controlled setting were effective and well-tolerated in reducing PTSD symptoms in veterans and first responders.
Enriched metadata from Blossom.
-
Ot'alora G, M., Grigsby, J., Poulter, B., Van DerVeer, J.W. 3rd., Giron, S.G., Jerome, L., Feduccia, A.A., Hamilton, S., Yazar-Klosinski, B., Emerson, A., Mithoefer, M.C., Doblin, R. (2018). 3,4-Methylenedioxymethamphetamine-assisted psychotherapy for treatment of chronic posttraumatic stress disorder: A randomized phase 2 controlled trial. J Psychopharmacol, 32(12), 1295-1307. https://doi.org/10.1177/0269881118806297
Study summary via Blossom
In this randomised phase 2 dose‑response trial, two active MDMA doses (100 mg and 125 mg) given with eight‑hour psychotherapy sessions produced larger reductions in clinician‑rated PTSD severity than a low (40 mg) dose. Benefits were sustained at 12‑month follow‑up (76% no longer met PTSD criteria) and the treatment was well tolerated with no drug‑related serious adverse events.
Abstract
Background
Posttraumatic stress disorder often does not resolve after conventional psychotherapies or pharmacotherapies. Pilot studies have reported that 3,4-methylenedioxymethamphetamine (MDMA) combined with psychotherapy reduces posttraumatic stress disorder symptoms.
Aims
This pilot dose response trial assessed efficacy and safety of MDMA-assisted psychotherapy across multiple therapy teams.
Methods
Twenty-eight people with chronic posttraumatic stress disorder were randomized in a double-blind dose response comparison of two active doses (100 and 125 mg) with a low dose (40 mg) of MDMA administered during eight-hour psychotherapy sessions. Change in the Clinician-Administered PTSD Scale total scores one month after two sessions of MDMA served as the primary outcome. Active dose groups had one additional open-label session; the low dose group crossed over for three open-label active dose sessions. A 12-month follow-up assessment occurred after the final MDMA session.
Results
In the intent-to-treat set, the active groups had the largest reduction in Clinician-Administered PTSD Scale total scores at the primary endpoint, with mean (standard deviation) changes of −26.3 (29.5) for 125 mg, −24.4 (24.2) for 100 mg, and −11.5 (21.2) for 40 mg, though statistical significance was reached only in the per protocol set ( p=0.03). Posttraumatic stress disorder symptoms remained lower than baseline at 12-month follow-up ( p<0.001) with 76% ( n=25) not meeting posttraumatic stress disorder criteria. There were no drug-related serious adverse events, and the treatment was well-tolerated.
Conclusions
Our findings support previous investigations of MDMA-assisted psychotherapy as an innovative, efficacious treatment for posttraumatic stress disorder.
Enriched metadata from Blossom.
-
Mitchell, J.M., Bogenschutz, M., Lilienstein, A., Harrison, C., Kleiman, S., Parker-Guilbert, K., Ot'alora G, M., Garas, W., Paleos, C., Gorman, I., Nicholas, C., Mithoefer, M., Carlin, S., Poulter, B., Mithoefer, A., Quevedo, S., Wells, G., Klaire, S.S., van der Kolk, B., Tzarfaty, K., Amiaz, R., Worthy, R., Shannon, S., Woolley, J.D., Marta, C., Gelfand, Y., Hapke, E., Amar, S., Wallach, Y., Brown, R., Hamilton, S., Wang, J.B., Coker, A., Matthews, R., de Boer, A., Yazar-Klosinski, B., Emerson, A., Doblin, R. (2021). MDMA-assisted therapy for severe PTSD: a randomized, double-blind, placebo-controlled phase 3 study. Nat Med, 27(6), 1025-1033. https://doi.org/10.1038/s41591-021-01336-3
Study summary via Blossom
In a randomized, double‑blind, placebo‑controlled phase 3 trial of 90 patients with severe PTSD (including common comorbidities), MDMA‑assisted therapy produced a large, significant reduction in PTSD symptoms versus placebo (mean CAPS‑5 change −24.4 vs −13.9; P<0.0001, d=0.91) and improved functioning (SDS: P=0.0116, d=0.43). Treatment was well tolerated with no signals of abuse potential, increased suicidality or QT prolongation, indicating MDMA‑assisted therapy is a promising intervention warranting expedited evaluation.
Abstract
Post-traumatic stress disorder (PTSD) presents a major public health problem for which currently available treatments are modestly effective. We report the findings of a randomized, double-blind, placebo-controlled, multi-site phase 3 clinical trial (NCT03537014) to test the efficacy and safety of 3,4-methylenedioxymethamphetamine (MDMA)-assisted therapy for the treatment of patients with severe PTSD, including those with common comorbidities such as dissociation, depression, a history of alcohol and substance use disorders, and childhood trauma. After psychiatric medication washout, participants ( n = 90) were randomized 1:1 to receive manualized therapy with MDMA or with placebo, combined with three preparatory and nine integrative therapy sessions. PTSD symptoms, measured with the Clinician-Administered PTSD Scale for DSM-5 (CAPS-5, the primary endpoint), and functional impairment, measured with the Sheehan Disability Scale (SDS, the secondary endpoint) were assessed at baseline and at 2 months after the last experimental session. Adverse events and suicidality were tracked throughout the study. MDMA was found to induce significant and robust attenuation in CAPS-5 score compared with placebo ( P < 0.0001, d = 0.91) and to significantly decrease the SDS total score ( P = 0.0116, d = 0.43). The mean change in CAPS-5 scores in participants completing treatment was −24.4 (s.d. 11.6) in the MDMA group and −13.9 (s.d. 11.5) in the placebo group. MDMA did not induce adverse events of abuse potential, suicidality or QT prolongation. These data indicate that, compared with manualized therapy with inactive placebo, MDMA-assisted therapy is highly efficacious in individuals with severe PTSD, and treatment is safe and well-tolerated, even in those with comorbidities. We conclude that MDMA-assisted therapy represents a potential breakthrough treatment that merits expedited clinical evaluation.
Enriched metadata from Blossom.
-
Mitchell, J.M., Ot'alora G, M., van der Kolk, B., Shannon, S., Bogenschutz, M., Gelfand, Y., Paleos, C., Nicholas, C.R., Quevedo, S., Balliett, B., Hamilton, S., Mithoefer, M., Kleiman, S., Parker-Guilbert, K., Tzarfaty, K., Harrison, C., de Boer, A., Doblin, R., Yazar-Klosinski, B. (2023). MDMA-assisted therapy for moderate to severe PTSD: a randomized, placebo-controlled phase 3 trial. Nat Med, 29(10), 2473-2480, https://doi.org/10.1038/s41591-023-02565-4
Study summary via Blossom
This multi-site, randomised, double-blind, Phase IIIb trial (n=104) evaluated the efficacy and safety of MDMA-assisted therapy (MDMA-AT) for individuals with moderate to severe PTSD. The study found significant reductions in PTSD severity (CAPS-5 score) and functional impairment (SDS score) for the MDMA-AT group compared to placebo with therapy. Seven participants experienced severe treatment-emergent adverse events, but no deaths or serious adverse events were reported. The treatment was found to be generally well tolerated in a diverse population.
Abstract
This multi-site, randomized, double-blind, confirmatory phase 3 study evaluated the efficacy and safety of 3,4-methylenedioxymethamphetamine-assisted therapy (MDMA-AT) versus placebo with identical therapy in participants with moderate to severe post-traumatic stress disorder (PTSD). Changes in Clinician-Administered PTSD Scale for DSM-5 (CAPS-5) total severity score (primary endpoint) and Sheehan Disability Scale (SDS) functional impairment score (key secondary endpoint) were assessed by blinded independent assessors. Participants were randomized to MDMA-AT (n = 53) or placebo with therapy (n = 51). Overall, 26.9% (28/104) of participants had moderate PTSD, and 73.1% (76/104) of participants had severe PTSD. Participants were ethnoracially diverse: 28 of 104 (26.9%) identified as Hispanic/Latino, and 35 of 104 (33.7%) identified as other than White. Least squares (LS) mean change in CAPS-5 score (95% confidence interval (CI)) was −23.7 (−26.94, −20.44) for MDMA-AT versus −14.8 (−18.28, −11.28) for placebo with therapy (P < 0.001, d = 0.7). LS mean change in SDS score (95% CI) was −3.3 (−4.03, −2.60) for MDMA-AT versus −2.1 (−2.89, −1.33) for placebo with therapy (P = 0.03, d = 0.4). Seven participants had a severe treatment emergent adverse event (TEAE) (MDMA-AT, n = 5 (9.4%); placebo with therapy, n = 2 (3.9%)). There were no deaths or serious TEAEs. These data suggest that MDMA-AT reduced PTSD symptoms and functional impairment in a diverse population with moderate to severe PTSD and was generally well tolerated.
Enriched metadata from Blossom.